You know the saying “little kids, little problems; big kids, big problems?” Anyone in the thick of parenting small children may question this saying, but it suggests that small children have problems that are simpler and more easily solvable than problems faced by older kids.
In compliance, this is not the case. Because regulation of life sciences companies is size agnostic, being a small or medium-sized company does not reduce the level or quality of the compliance you need to achieve.
A recent study we conducted on how a digital CAPA solution is implemented across over 80 life-science companies of various sizes and stages finds that this isn’t just a gut feeling. Our findings clearly show that small companies – working with far less resources than enterprise-level companies- need to maintain the same caliber of excellence, which is based on the ability to make highly specific processes aligned to their needs.
Level of Customization
Large life sciences companies make an average of 42 changes to CAPA best practices, adapting out-of-the-box practices to meet their specific needs. Interestingly, however, companies one-fifth of the size, make an average of 37 changes, indicating that the need for customization is not a function of company size. Based on our analysis, small companies don’t stop at best practices and engage in significant tailoring of those practices to their unique needs.

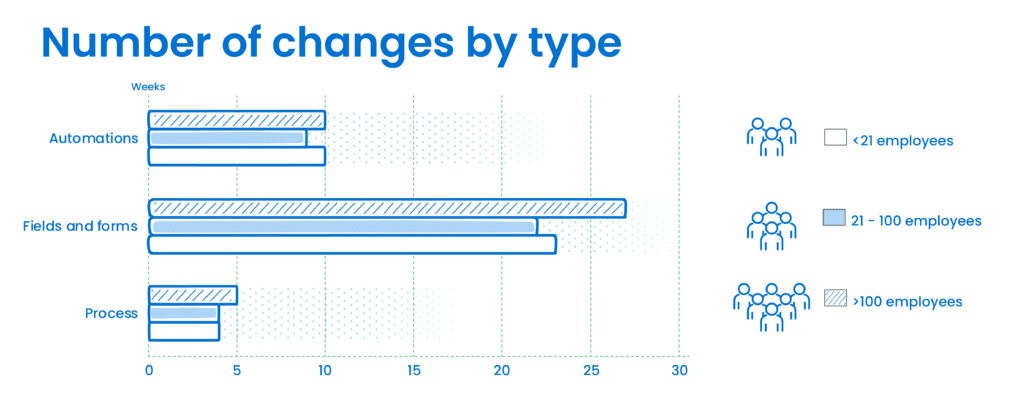
Below is a further drill down into the customization selected by type of change and company size. While large companies make more changes to fields and formats, small and medium size companies make a similar number of such changes, and all three groups make a similar number of edits to automations and process in order to meet their needs.
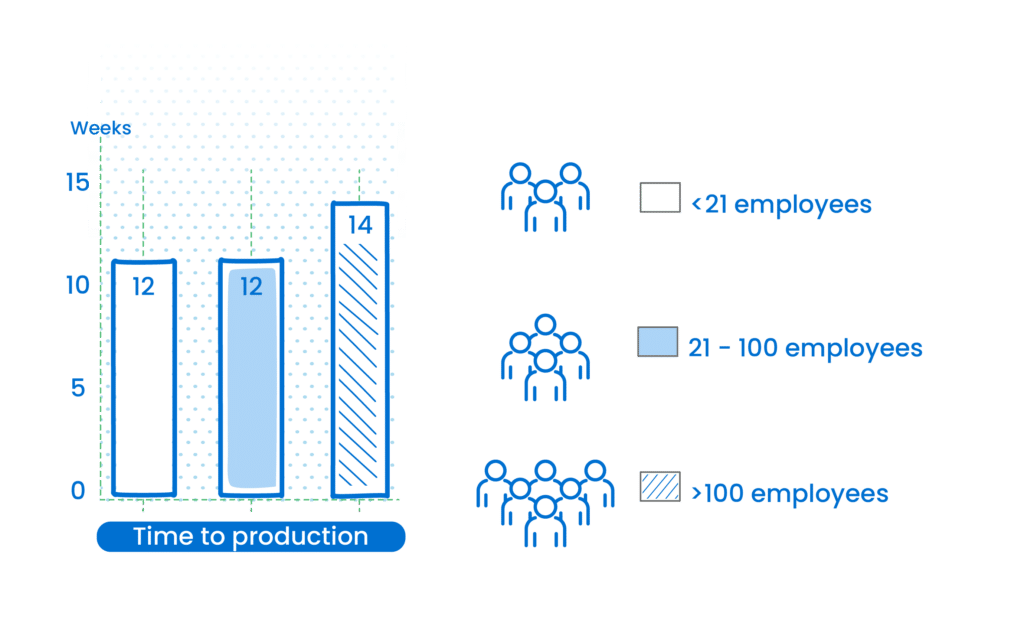
Time to Production
The journey from concept to production varies less than expected when comparing small businesses to large enterprises. While large companies take an average of 14 weeks, both small and medium-sized companies take 12 weeks – another indication that size doesn’t always dictate speed or efficiency in the market.
Data Migration:
Often perceived as a Herculean task, the time required for data migration didn’t significantly differ between small and large companies (An average of 8 weeks for both large and small companies; 7 weeks for medium sized companies), indicating that the phases of data preparation, correction, and migration tend to be consistent. And while the amount of data is typically larger in bigger, longer-operating businesses, the amount of files doesn’t have a significant impact on timeliness.
Taken together, this analysis finds that small and medium-sized companies are meeting the same level of nuance and specification in their compliance efforts as mature life science enterprises, despite massively reduced resources. Under the hood are teams that are highly committed to compliance excellence and technology that enables small companies to go well beyond out-of- the-box solutions. This is a critically important levelling of the playing field, as it allows small companies to think – and act – well beyond one-size-fits-all best practices.